|
The difference between the atom's number of valence electrons and the number it owns is the formal charge. Count all of its lone pair electrons, and half of its bonding electrons. Apart from the formula, the formal charge can be calculated pictorially using the diagrammatic method. This suggests that option A is the correct answer. To find formal charges in a Lewis structure, for each atom, you should count how many electrons it 'owns'. The formal charges on the nitrogen atoms in the case of azide ion from left to right are -1, +1, -1. Hence the central nitrogen atom of the azide ion has a +1 formal charge. The formal charge on the central nitrogen atom = The bonding electrons and nonbonding electrons in the central nitrogen atom of azide are eight and zero, respectively. The valence electrons in the N atom are five. This suggests that nitrogen atoms at the terminal positions of azide ion have a -1 formal charge.Ĭalculation of formal charge for the central nitrogen atom of azide ion. Putting these values in the above equation.įormal charge on terminal nitrogen atoms = -1. The bonding and nonbonding electrons in the terminal nitrogen atom of the azide ion are four. When calculating formal charge - electrons are shared equally betweenthe atoms in the bond.When calculating oxidation state - electrons are both given to. Hence N has five electrons in the valence shell. As a result chlorine is a negative charged ion. Once the chlorine gains an electron, however, the total just becomes -1 since there are now 18 electrons and still 17 protons. The electronic configuration of the N atom is 1s² 2s² 2p³. Chlorine has 17 electrons (a charge of -17) and 17 protons (a charge of +17), so the overall charge is zero. This suggests that the formal charge on these nitrogen atoms will also same. The terminal nitrogen atoms of the azide ion have the same chemical environment. The geometry of Molecules is a platform where we try to educate you about various chemistry concepts and find the answers you have been looking for.Formal charge = (number of valence electrons) - (number of nonbonding electrons) - (number of bonding electrons) 2 \textįirst, calculate the formal charge on terminal Nitrogen atoms of azide ion. And it might seem challenging, but it becomes the most fun and easy subject to learn if learned the correct way. Sounds easy, right? Go ahead and try our formal charge calculator.ĭo you know that a perfect diamond is made up of a single Carbon molecule? Chemistry is one of the most exciting subjects. Solution: Formal charge can be calculated by using the formula: FC V - N - B/2 Where, FC formal charge V number of valence electrons N number of. So, for as long as you have a complete Lewis structure and periodic table handy, you can quickly find the formal charge of any atom in a molecule. 
And to make it easy for you to do the calculations we have developed a Formal charge calculator for you, where you have to just enter the number of valence electrons, non-bonding electrons, and bonding electrons to find out the formal charge. Holidays and weekends are included in the calculation. Similarly, you can use this formula for other molecules and find out formal charges for individual atoms in the molecule. Other forums for resolution may be pursued at the same time as the processing of the EEOC charge. And using the formula stated above, we found out that the total charges on both Sulphur and Fluorine atoms are zero. Here in SF4 as all the Fluorine atoms are arranged symmetrically, they will have the same charges. 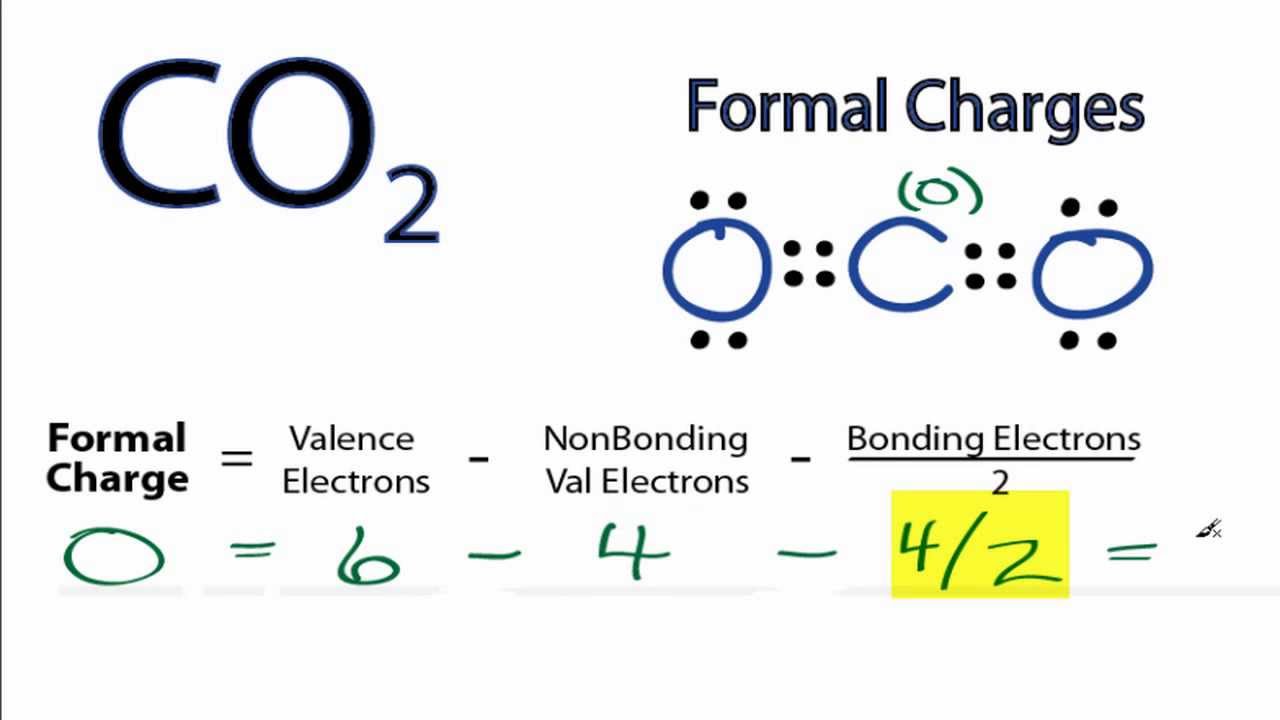
of valence electrons Non-bonding electrons Bonding electrons Total charge Sulphur (S) About Press Copyright Contact us Creators Advertise Developers Terms Privacy Policy & Safety How YouTube works Test new features Press Copyright Contact us Creators. Now that we know the formula let us look at the example of how to find out formal charges for individual atoms in a polyatomic molecule Let us calculate the formal charges of SF4 Atom Total no. One can calculate the formal charges for any given atom with the help of the following formula:į.C = Valence electrons – Nonbonding electrons- Bonding electrons/2 The formal charge of an atom in a molecule is the hypothetical charge the atom would have if we could redistribute the electrons in the bonds evenly between the atoms. These charges help in knowing if the given structure of the molecule is stable or not. 
Formal charges are important because they allow us to predict which Lewis structure is the mo. Formal charge is the individual electric charges on the atoms in a given polyatomic molecule. A step-by-step description on how to calculate formal charges.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
